-
See Also
- IsotopeData
- ChemicalData
- ParticleData
- LatticeData
- SpectralLineData
-
- Entity Types
- Element
-
- Interpreter Types
- Element
- ElementClass
- ComputedElement
- Related Guides
-
-
See Also
- IsotopeData
- ChemicalData
- ParticleData
- LatticeData
- SpectralLineData
-
- Entity Types
- Element
-
- Interpreter Types
- Element
- ElementClass
- ComputedElement
- Related Guides
-
See Also
ElementData["name","property"]
gives the value of the specified property for the chemical element "name".
ElementData[n,"property"]
gives the specified property for the n![]() chemical element.
chemical element.


ElementData
ElementData["name","property"]
gives the value of the specified property for the chemical element "name".
ElementData[n,"property"]
gives the specified property for the n![]() chemical element.
chemical element.
Details











- Chemical elements can be specified by their full names, standard abbreviations, or atomic numbers.
- ElementData[] gives a list of all standard chemical elements.
- ElementData[n] gives the full name of the n
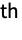 chemical element.
chemical element. - ElementData["Properties"] gives a list of all properties available for chemical elements.
- Properties that do not apply or are not known in a particular case are indicated by Missing[…].
- Numerical values that are known only to a certain precision are typically tagged with their precision and given in arbitrary-precision form.
- Basic element properties include:
-
"AtomicNumber" atomic number 
"AtomicWeight" average atomic weight in atomic mass units - If no other temperature or pressure is specified, all macroscopic properties are given for standard temperature and pressure. When applicable, properties are for the most stable form of the element at standard temperature and pressure.
- Material properties include:
-
"BrinellHardness" Brinell indentation hardness "BulkModulus" bulk modulus (incompressibility) 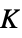
"Density" density at standard temperature and pressure "LiquidDensity" liquid density at melting point "MohsHardness" Mohs scratch resistance "MolarVolume" molar volume "PoissonRatio" Poisson ratio 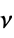 of solid
of solid"ShearModulus" shear modulus 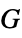 of solid
of solid "SoundSpeed" speed of sound "ThermalConductivity" thermal conductivity 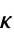
"ThermalExpansion" linear thermal expansion coefficient 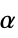
"VickersHardness" Vickers indentation hardness "YoungModulus" Young's modulus 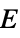 of solid
of solid - Thermodynamic properties include:
-
"AbsoluteBoilingPoint" boiling temperature in kelvin "AbsoluteMeltingPoint" melting temperature in kelvin "AdiabaticIndex" gas phase adiabatic index 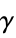
"BoilingPoint" boiling temperature in degrees Celsius "CriticalPressure" pressure at liquid-gas critical point "CriticalTemperature" temperature at liquid-gas critical point "CuriePoint" Curie temperature at ferromagnet-paramagnet transition "FusionHeat" latent heat for solid-liquid transition "MeltingPoint" melting temperature in degrees Celsius "NeelPoint" Néel temperature at antiferromagnet-paramagnet transition "Phase" naturally occurring phase (e.g. "Solid") "SpecificHeat" specific heat capacity 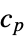
"SuperconductingPoint" superconducting transition temperature in kelvin "VaporizationHeat" latent heat for liquid-gas transition - Electromagnetic and optical properties include:
-
"Color" basic material color "ElectricalConductivity" electrical conductivity 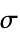
"ElectricalType" electrical type (e.g. "Insulator") "MagneticType" magnetic response type (e.g. "Paramagnetic") "MassMagneticSusceptibility" mass magnetic susceptibility 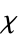
"MolarMagneticSusceptibility" molar magnetic susceptibility 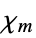
"RefractiveIndex" refractive index for visible light "Resistivity" electrical resistivity 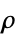 of solid
of solid"VolumeMagneticSusceptibility" volume magnetic susceptibility 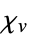
- Abundance properties include (specified by mass-fraction):
-
"CrustAbundance" Earth's crust abundance "HumanAbundance" typical human body abundance "MeteoriteAbundance" observed carbonaceous meteorite abundance "OceanAbundance" ocean abundance "SolarAbundance" abundance in the Sun "UniverseAbundance" observed universe abundance - Periodic table properties include:
-
"Block" label of outermost occupied electron subshell "Group" column in periodic table "Period" row in periodic table (number of electron shells) "Series" named series of elements (e.g. "AlkaliMetals") - Basic chemical properties include:
-
"AllotropicMultiplicities" numbers of atoms in allotropic molecules "CommonCompoundNames" names of common compounds "ElectronAffinity" electron affinity "Electronegativity" Pauling electronegativity "GasAtomicMultiplicities" number of atoms in gas-phase molecules "Valence" number of chemical bonds typically formed - Crystallographic properties for the most stable solid phase include:
-
"CrystalStructure" basic crystal lattice structure "LatticeAngles" angles between crystallographic axes 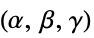
"LatticeConstants" crystal lattice constants 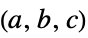
"SpaceGroupNumber" International Tables space group number "SpaceGroup" space group symbol - Atomic properties include:
-
"AtomicRadius" calculated distance from nucleus of outermost electron "CovalentRadius" half-distance between nuclei in a bonded pair of atoms "ElectronConfiguration" occupation numbers for each electron subshell "ElectronConfigurationString" electron configuration written in standard notation "ElectronShellConfiguration" occupation numbers for each complete energy shell "IonizationEnergies" list of ionization energies for each electron "QuantumNumbers" ground state angular momentum quantum numbers "VanDerWaalsRadius" hard sphere interatomic interaction range - Nuclear properties include:
-
"DecayMode" dominant decay mode for longest-lived isotope "IsotopeAbundances" list of mass numbers and abundances of stable isotopes "IsotopeHalfLives" half-lives for all known isotopes "IsotopeLifetimes" exponential lifetimes for all known isotopes "KnownIsotopes" atomic mass numbers of all known isotopes "MolarRadioactivity" radioactivity per mole "NeutronCrossSection" thermal neutron cross section in barns "NeutronMassAbsorption" thermal neutron mass absorption coefficient "SpecificRadioactivity" specific radioactivity "StableIsotopes" atomic mass numbers of stable isotopes - Names-related properties include:
-
"Abbreviation" standard element abbreviation (e.g. "Na") "AllotropeNames" names of allotropes (e.g. "Graphite") "AlternateNames" alternate names (e.g. "Aluminium") "CASNumber" Chemical Abstract Service registry number "IconColor" icon color for Wolfram Language molecular graphics "Name" English name (e.g. "hydrogen") "StandardName" standard Wolfram Language name (e.g. "Hydrogen") - Historical and commercial properties include:
-
"DiscoveryCountries" countries where element was discovered "DiscoveryYear" year element was discovered - ElementData["class"] gives a list of elements in the specified class. ElementData[name,"class"] gives True or False depending on whether the element corresponding to name is in the specified class.
- Classes of elements include:
-
"Actinide" actinide "AlkaliMetal" alkali metal "Gas" gas at standard temperature and pressure "Lanthanide" lanthanide "Liquid" liquid at standard temperature and pressure "Ferromagnetic" ferromagnetic at standard temperature and pressure "Metal" metallic "Metalloid" metalloid "Natural" naturally occurring "Nonmetal" non-metallic "NobleGas" noble gas "Radioactive" radioactive in all forms "Solid" solid at standard temperature and pressure "Stable" stable with respect to radioactive decay "Synthetic" occurring only when synthesized - ElementData["name","property","ann"] gives various annotations associated with a property. Typical annotations include:
-
"Description" short textual description of the property "Interval" uncertainty range given by Interval[…] "LongDescription" longer textual description of the property "Note" additional information about the property "Units" units in which the property is given "UnitsName" English name for the units used "UnitsNotation" notation for the units used "UnitsStandardName" Wolfram Language standard name for the units used "Value" property value (default if no annotation is specified)
Examples
open all close allBasic Examples (3)
Scope (17)
Names and Classes (7)
Obtain a list of all the elements sorted by atomic number:
Find the English name of an element:
A list of alternate names can also be found:
An element can be specified by its standard Wolfram Language name, atomic number, or abbreviation:
Additional names acceptable as input can be found:
Find the list of element classes:
Properties and Annotations (5)
Get a list of properties for a particular element:
Get a short textual description of a property:
Get a longer textual description:
Find the units in which a property value is given:
Get units in proper textual form:
Uncertainty is represented using Interval:
Property Values (5)
A property value can be any valid Wolfram Language expression:
Use Precision to find out the number of significant digits for a property:
A property that is not applicable to an element has the value Missing["NotApplicable"]:
A property that is not available for an element has the value Missing["NotAvailable"]:
A property that is unknown for an element has the value Missing["Unknown"]:
Applications (6)
Show the linear relationship between enthalpy of vaporization and boiling point:
Plot the atomic radii of the elements, showing the effects of the electron shell structure:
Plot the number of stable isotopes of each element:
Plot the atomic masses of all stable isotopes:
Create an annotated list of critical temperature and critical pressure:
Use the description annotations to get plot labels:
Draw a basic periodic table, color coding solids, liquids, and gases:
Properties & Relations (9)
Use Table to obtain a list of property values for all the elements:
The same thing using Map:
Form a list of property values for a class of elements:
Display the data as a formatted table using Grid:
Use DeleteCases to filter out non-missing elements:
Use ListPlot to plot a list of property values:
Use ListLogPlot when the data spans several orders of magnitude:
Use LatticeData to get detailed information on solid state structures:
Use IsotopeData to get detailed information on isotopes:
Use ChemicalData to find properties for compounds of elements:
Possible Issues (2)
See Also
IsotopeData ChemicalData ParticleData LatticeData SpectralLineData
Entity Types: Element
Interpreter Types: Element ElementClass ComputedElement
Function Repository: AtomicNumber FromAtomicNumber
Related Guides
Related Links
History
Introduced in 2007 (6.0) | Updated in 2008 (7.0) ▪ 2014 (10.0)
Text
Wolfram Research (2007), ElementData, Wolfram Language function, https://reference.wolfram.com/language/ref/ElementData.html (updated 2014).
CMS
Wolfram Language. 2007. "ElementData." Wolfram Language & System Documentation Center. Wolfram Research. Last Modified 2014. https://reference.wolfram.com/language/ref/ElementData.html.
APA
Wolfram Language. (2007). ElementData. Wolfram Language & System Documentation Center. Retrieved from https://reference.wolfram.com/language/ref/ElementData.html
BibTeX
@misc{reference.wolfram_2025_elementdata, author="Wolfram Research", title="{ElementData}", year="2014", howpublished="\url{https://reference.wolfram.com/language/ref/ElementData.html}", note=[Accessed: 08-May-2026]}
BibLaTeX
@online{reference.wolfram_2025_elementdata, organization={Wolfram Research}, title={ElementData}, year={2014}, url={https://reference.wolfram.com/language/ref/ElementData.html}, note=[Accessed: 08-May-2026]}